Experiments This Week
Monday – setup of ligation reaction with the new rtcB and substrates; calculated amount of RNA and hybrid stocks needed, recalculated all the substrate conc; calculated amount, reaction conditions and parameters for positive control; looked up ethanol precipitation. Performed ligation reaction in 37C water bath overnight.
Tuesday – Attempted to pour two 18% SDS-PAGE RNA gels, both of which failed, due to detachment from the glass resulting in bubbles. Performed the T4PNK and Shrimp Phosphatase reactions for the positive control. Let the tubes air dry with a kimwipe on top, overnight. Performed second ligation reaction in 37C water bath overnight.
Wednesday – Attempted to pour two 18% SDS-PAGE RNA gels, using the TEMED and APS amounts recommended in Biorad tech note, as well as the 30% Acrylamide/Bis (37.5:1) ratio, for DNA. Usually, I’d been using the (29:1) ratio for protein, because it didn’t seem to have any effect. Looked up notes on Biorad about polyacrylamide gels, checked expiration date of supplies; confirmed expiration date of newly ordered 5g Ampicillin from Sigma-Aldrich.
The RNA gels failed due to detachment from the glass and bubbles. One was salvageable. Ordered 3 sets of two 15% precast gels from Biorad. Walked to Whitehead and bought a set of 4%-20% gradient Tris-HCl gels, to see if I could get separation with my ligation reaction.
Heated ligation reaction samples at 65C 10 min. Ran them on the gel ~45 min. Imaged on Typhoon, but the results were inconclusive. Checked the amount of DNA/RNA substrate in my positive control tubes on Nanodrop. It was there, but pretty low, so I didn’t end up doing the ligation reaction. Need to optimize the setup and/or the ethanol precipitation procedure.
Thursday – Poured two 18% RNA gels with the 30% Acrylamide/Bis (37.5:1) ratio, as previously. Setup a third ligation reaction, w/ substrate conc tweaked for better loading.
Friday – The ligation reaction waterbath was shut off prematurely but seems not to have effected the results. Poured two 18% gels using 10-well combs, since seemed to get fewer bubbles when used those previously. Yoshie thought it should run even with the detatchment. Bubbles appeared but mostly around sides. Ran the gels of ligation rxn, stained hybrid gel w/ SYBR gold, imaged on Typhoon. No serious effect of the bubbles was noted.
Many lanes missing in the gels; however, the hybrid substrate gel shows what appears to be product in the “rxn, rtcB” lane that matches the band in the 20mer lane. RNA FAM gel is inconclusive and seems to have suffered from degradation.
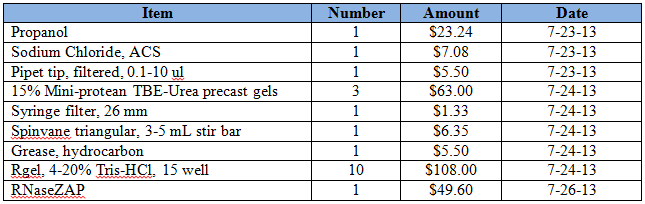
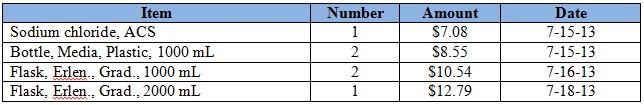
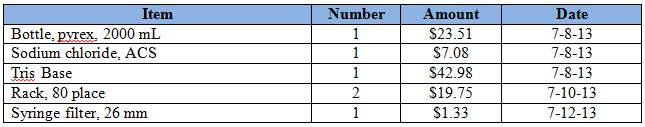
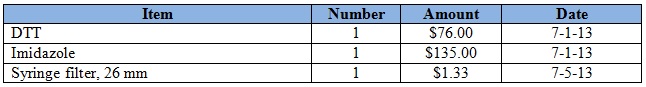
Purchases for the week = $269.60

 RSS Feed
RSS Feed
