CHAPTER 16 – STABILITY OF AROMATIC CMDS
1. (16-1, 2) Why is benzene so stable? There are 2 reasons – can you come up with both?
a. Resonance
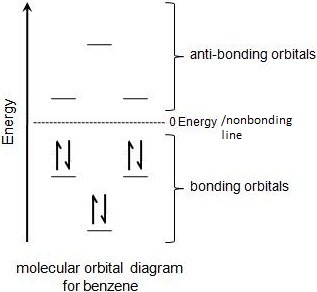
b. Even more importantly, it has all filled bonding molecular orbitals (MOs) – and none of the anti-bonding MOs are filled
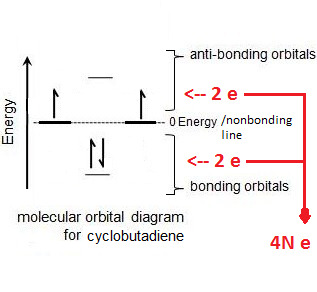
§ NOTE: If benzene was only stable due to resonance around the ring, we would expect cyclobutadiene (an anti-aromatic compound) to also be as stable
§ NOTE: This is what figure 16-2 and the end of section 16-2 in the book is showing
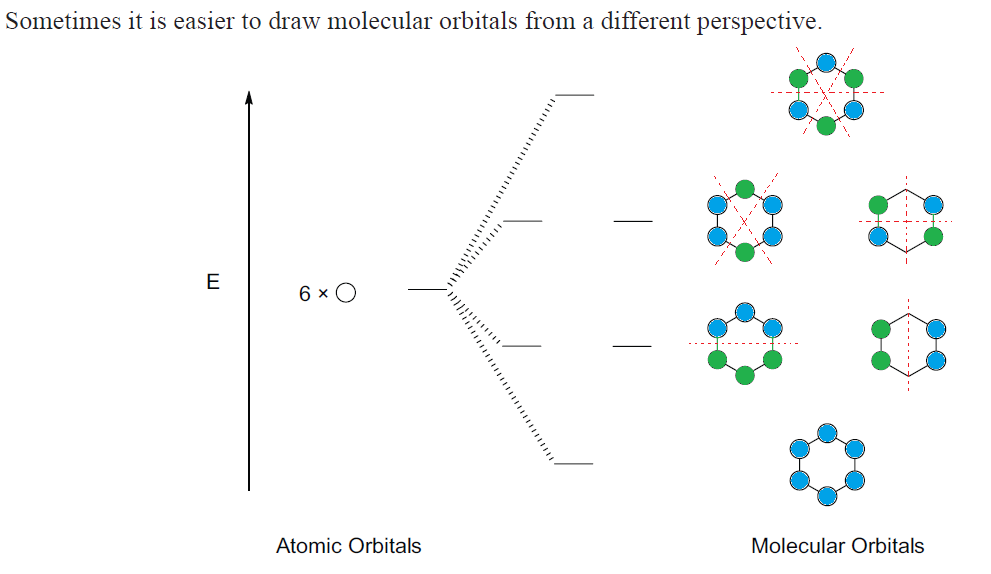
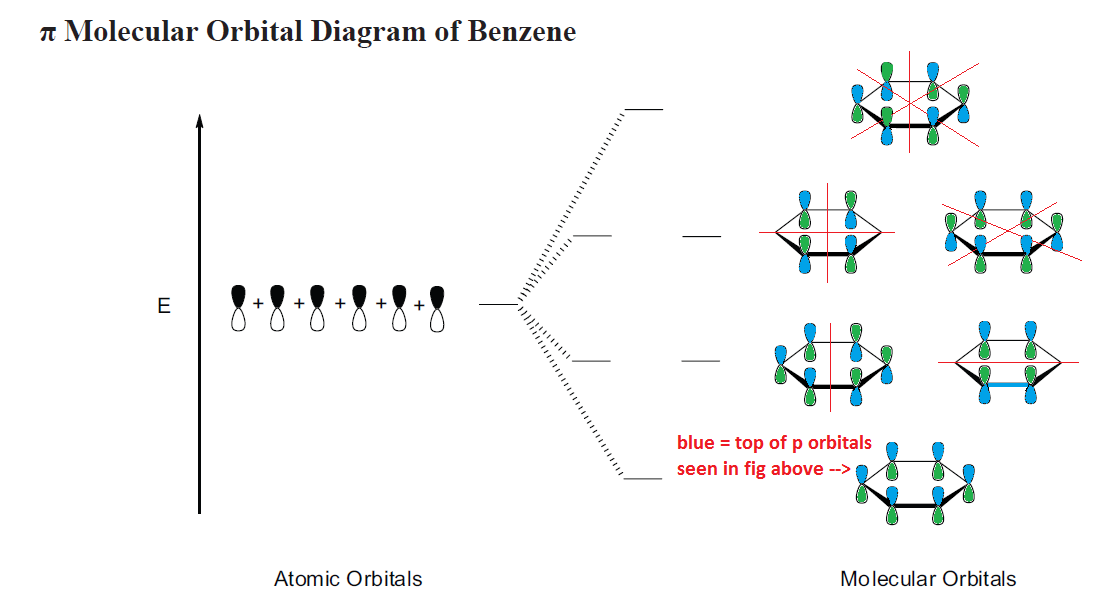
2. Molecular orbitals of benzene
a. The textbook shows benzene’s molecular orbitals similar to this (see Fig. 16-4) Note the following….
b. The different colors represent the different phases of the p orbitals, just as in the text
c. This is a top down view, looking at the top of benzene’s ring – thus, you can only see the top of the p orbitals
d. Below the ring, the bottom p orbitals have the opposite phase from the top ones, as you’d expect
e. Nodes are indicated by a red line
f. “E” on the left, refers to energy. The lowest orbital is all bonding (shown w/ the tops of all p orbitals the same phase –blue) and also the lowest energy and most stable.
g. The 2nd and 3rd levels of orbitals are of equal energy – thus, they are referred to as “degenerate orbitals”
h. The electron configuration is drawn according to the polygon rule
i. Only two electrons can fit into each MO configuration
j. When a node falls on top of a carbon, there is zero electron density on that carbon. Thus, the p orbital is still there, but it is empty, so it is not shown.
1. The Polygon Rule states: the electron configuration of a polygon (cyclic, conjugated) is the same shape as the polygon, with one vertex at the bottom (corresponding to the all-bonding molecular orbital)
2. The second level of orbitals has 1 node, with a node being added each level
3. If one counts the bonding, anti-bonding and non-bonding interactions of each MO, you can see that each pair of degenerate orbitals have the same number of net interactions (which is why they are of = energy)
a. Interactions are measured from carbon to neighboring carbon
b. Bonding interactions – no node between a carbon and its neighbor (see 1st level of orbitals; top two blue orbitals on right, 2nd level, etc)
c. Anti-bonding interactions – there is a node between a carbon and its neighbor (see 2nd level, left, top two orbitals – green and blue)
d. Non-bonding interactions – occurs when the carbon adjacent to another carbon is on the node (see 2nd level, right – node is on the middle carbons)
e. Anti-bonding and bonding interactions cancel each other, while non-bonding interactions don’t add or subtract anything
4. You cannot draw MOs the same way for cyclic compounds as for the linear compounds in ch. 15 (e.g., butadiene) – they must be drawn as shown above, with the cyclic ring included
Rules of Aromaticity:
*To be aromatic, a compound must follow the rules below…
1. It must be cyclic
2. It must be planar
3. It must have conjugation all around the ring
4. It must follow Huckel’s Rule (4N + 1)
Aromatic compounds – follow all four rules and are very stable
Anti-aromatic compounds – follow the first 3 rules, break the 4th, and are especially UN-stable
Non-aromatic compounds – break one of the first three rules (usually #3), and are of normal stability, not particular stable or unstable
Why Does Huckel’s Rule work? What does it mean?
**Huckel’s Rule is really just a mathematical shortcut to see if a polygon will have electrons in an all-bonding arrangement that’s really stable (like benzene) or an unstable, radical arrangement (like cyclobutadiene) – what do I mean by that? See below:
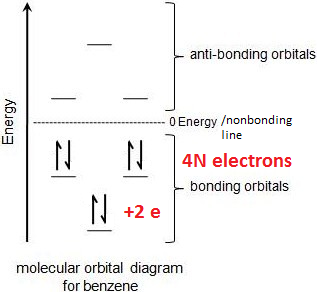
Benzene’s electron configuration:
3 double bonds in benzene = 2 e- per bond x 3 = 6 e-
1 p orbital per e- = 6 p orbitals
1 MO per p orbital = 6 MOs
What the Huckel rule is saying is, as long as you have 4N + 2 electrons, you will get a stable electron configuration.
HOWEVER, if you have 4N electrons, there won’t be enough to fill the first pair of degenerate orbitals, which leaves radicals – SEE BELOW
*First check to see if a compound follows the first 3 rules – if it does, check Huckel’s rule to determine if it’s aromatic or anti-aromatic. If it doesn’t, it’s non-aromatic.
Annulenes – a term used to quickly name any cyclic conjugated compound. The number of carbons in the compound are listed in brackets in front; thus, benzene = [6]annulene. It’s especially useful for naming very large rings.
Rules for large rings…
**Note: For determining whether or not large rings are planar… for annulenes 10 or greater, one can first check Huckel’s rule – if the ring has 4N + 2 electrons, then it WILL BE planar and thus aromatic. If it does not have 4N + 2 electrons, then it WILL NOT be planar and thus, non-aromatic.
**This is true, because annulenes 10 or greater have the flexibility to twist out of shape to avoid unfavorable conjugation, if it is anti-aromatic and vice versa. For annulenes less than 10, check the 1st three rules first, as usual.
**WARNING: Do not use this on bicyclic compounds – i.e., fused rings. “Annulenes 10 or greater” assumes that the carbons are all a part of one ring. Fused rings follow the usual order of rule checking.
**Tip: Just because an ion is aromatic, does not immediately imply that it is JUST AS STABLE AS BENZENE. It means that it’s much more stable than the equivalent non-conjugated or non-cyclic compound.
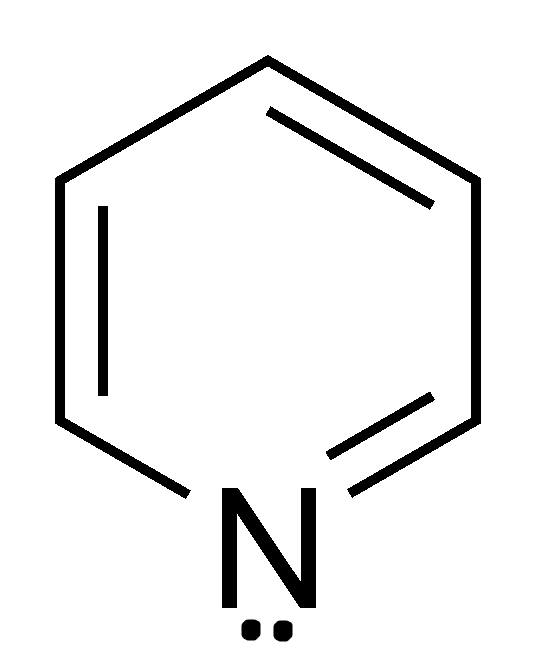
N: Basic or non-basic? That is the question!
*You will need to distinguish between basic and non-basic nitrogen. The examples for these are pyridine (containing a basic N) and pyrrole (w/ non-basic N).
1. The lone electron pairs on N are BASIC, because they are NOT needed for the conjugation inside the ring to be complete – note that there are already a set of alternating double bonds inside the ring.
2. Thus, the lone pairs are in an un-hybridized p orbital not attached to the conjugated ring system
3. This leaves them free to be donated to other compounds in reactions, thus making the N basic (Lewis definition).
Remember: A Lewis base donates electrons and a Lewis acid accepts electrons.
A Bronsted base accepts an H+ and a Bronsted acid donates an H+.
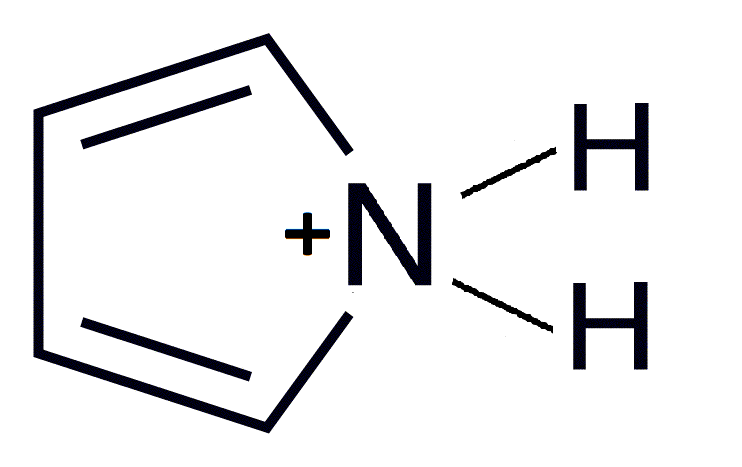
NOTE: For pyrrole
1. The lone electron pairs on pyrrole’s N are NON-BASIC, because they ARE needed for the conjugation inside the ring to be complete – there are only two double bonds and the bottom has none.
2. Thus, the lone pairs on N are in an sp2 orbital that is attached to the conjugated ring system
3. This makes them part of the aromatic ring, and thus it is not favorable for them to react with other compounds, because if they leave, the ring will no longer be aromatic.
4. Thus, they must be non-basic (Lewis definition).
CONSIDER:
*Nitrogen forms at max 4 bonds and prefers to form 3…
- Non-basic N’s tend to look like (2) – they have all single bonds attached to the N, and usually a H also
- If you see (3), that N has 4 bonds and is sp3 hybridized – thus it will break conjugation inside a ring and is non-basic by default, since it has no lone pair e- left to give
EXAMPLE:
If you see this – note, that the N has four bonds. There are no lone pairs and the N is sp3 hybridized. Therefore, this compound does not have conjugation around the ring (breaking rule 3) and is thus non-aromatic.
Do not be fooled by how the compound is drawn –
· if there is no (+) charge drawn on a N w/ 4 bonds, it is still non-basic, sp3 hybridized
· if there is no lone pair drawn or if the lone pair is drawn inside the ring on a basic nitrogen – beware! Look for the number of bonds N has and whether or not its lone pair is involved in ring conjugation or not.
Compounds to Remember!
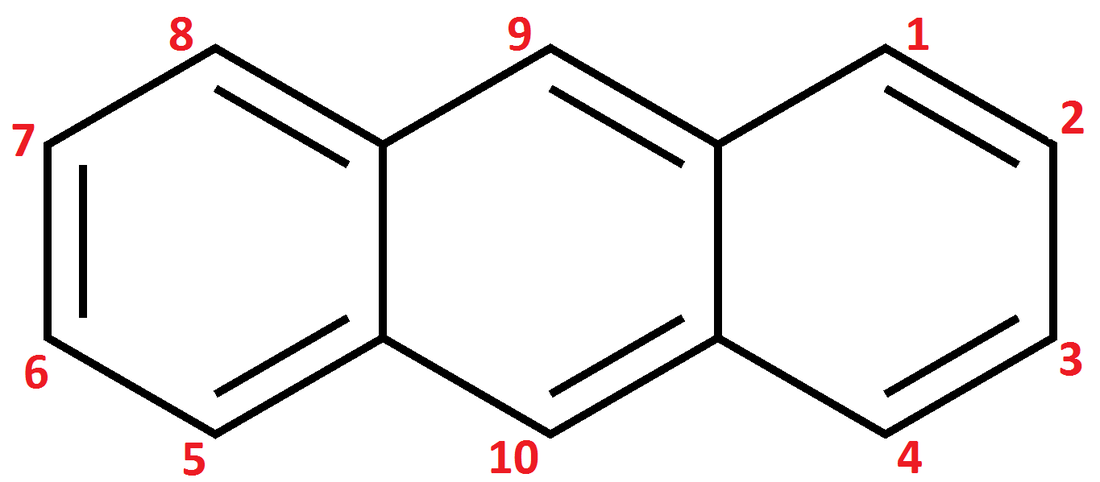
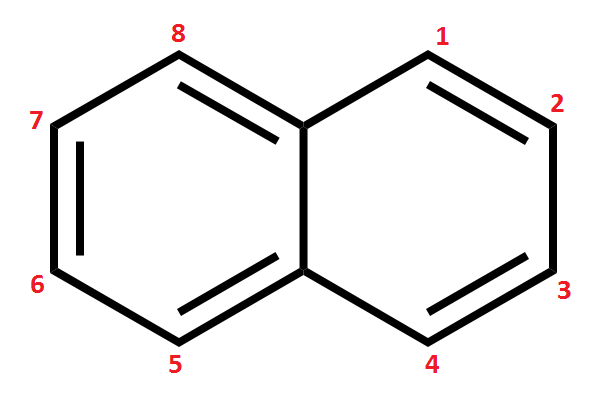
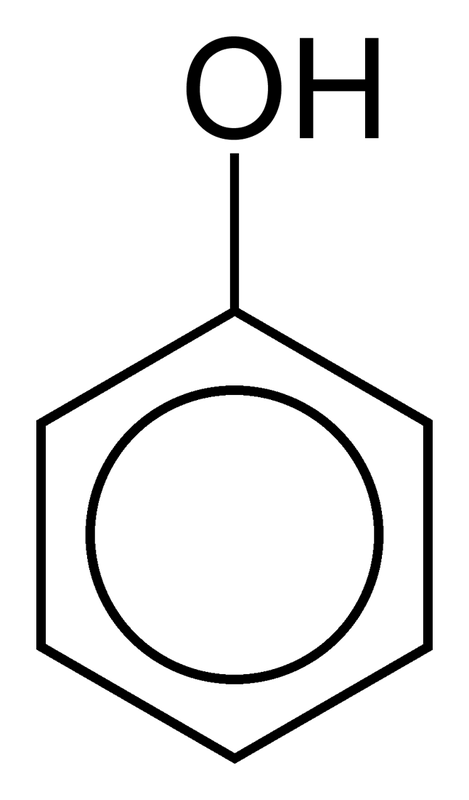
| anthracene (sect.16-10) **Remember to know how to number it **Note: the C’s where the rings fuse are NOT numbered – this is because these C’s never have substitutions on them in rxns. Numbering is largely for the benefit of labeling substitution products. This is also true for phenanthrene. Do not let it confuse you. | phenanthrene **Remember to know how to number it **Watch out for unlabeled C’s, same as above | naphthalene (moth balls) | phenol (sect 16-13) |
1. Phenyl group (Ph) – six carbon benzene ring attached to something
2. Benzyl group – seven carbon benzene ring + methylene attached to something
3. Aryl group(Ar) – Generic term for any aromatic group, just as generic alkyl groups are symbolized by “R”
**Fun Fact:
1. Pyridine is actually pronounced – “PEER-a-deen” http://www.merriam-webster.com/dictionary/pyridine
2. Pyrrole is pronounced – “PEER-roll” http://www.merriam-webster.com/dictionary/pyrrole
If you have any questions on this material, please let me know asap! Thanks! :D
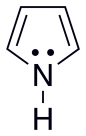
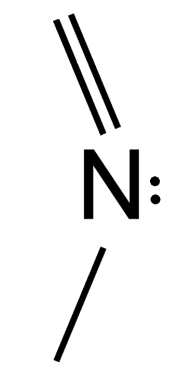









 RSS Feed
RSS Feed
