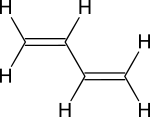
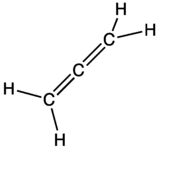
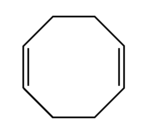
15-1 and 15-2 Stability of Dienes
· Understand difference between isolated double bonds, conjugated double bonds and cumulated double bonds and how each affects a compounds stability.
· See Fig 15-1. It is useful for comparing energies
· SEE PROBLEM: 15-1
o NOTE: In the problem, there is an error. In (b), the first structure has 3 double bonds. It should have only 2. The double bond on the bottom left should be crossed out.
15-3 Molecular Orbital Picture of a Conjugated System
· Resonance energy = the extra stability given by a conjugation in a molecule
· Molecular orbitals = current theory to explain where the extra stability due to conjugation comes from
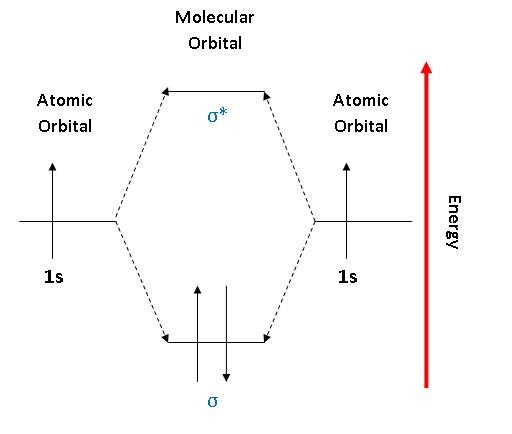
· Example: in 1,3-butadiene, each carbon has sp2 hybridized, overlapping p orbitals
· The p orbitals have two lobes; each lobe is opposite in the phase of its wave function
o +/- signs, as well as different colors are used to indicate this in text
o Orbitals of opposite phases cancel each other out; think troughs and crests having destructive interference, like in physics, except opposite phased orbitals cancelling would be in 3D
· Since they are conjugated, these overlapping p orbitals combine to make one big molecular orbital (MO), because of resonance. The electrons are delocalized over the whole molecule.
· The number of molecular orbitals a molecule has = the number of p orbitals that combined to make it. Incidentally, this is always going to be an even number, because you will always have resonance between pairs of carbons.
· Half of a molecule’s molecular orbitals will be bonding orbitals (the majority of the p orbitals interact constructively – most stable) and half will be anti-bonding (the majority of the p orbitals interact destructively – less stable)
· Electrons enter MOs in order of stability – most stable first, least stable last.
· Bonding MOs have lower energy than in the original p orbitals, and are preferred. Antibonding MOs have higher energy than the original p orbitals and are not preferred.
· In a molecule with two double bonds in resonance, there are four delocalized electrons, four p orbitals, thus four MOs. Two of those MOs are bonding, two are antibonding. Thus, two electrons enter the 1st molecular bonding orbital and two electrons enter the 2nd bonding molecular orbital. The anti-bonding orbitals are empty. This creates the most stable arrangement.
ONLINE MATERIAL RELATED TO SECTIONS 15-3, 15-8, 15-13 ON MOLECULAR ORBITALS
1. π Molecular Orbitals of Ethene: http://www.chem.ucalgary.ca/courses/351/Carey5th/Ch10/ch10-6-1.html
2. Molecular Orbital Theory: http://www.chemistryexplained.com/Ma-Na/Molecular-Orbital-Theory.html#b
3. Molecular Orbital Theory – thought it explained the interference concept pretty well: http://www.grandinetti.org/Teaching/Chem121/Lectures/MOTheory
4. Molecular Orbital Theory – another perspective: http://chemwiki.ucdavis.edu/Theoretical_Chemistry/Chemical_Bonding/Molecular_Orbital_Theory
15-4: Allylic Cations
· The allylic position is anything bonded to a carbon that is next to a C=C double bond
· Allylic cations are more stable than substituted cations, because they are next to a double bond and thus stabilized by resonance
· See problems 15-4 and 15-5
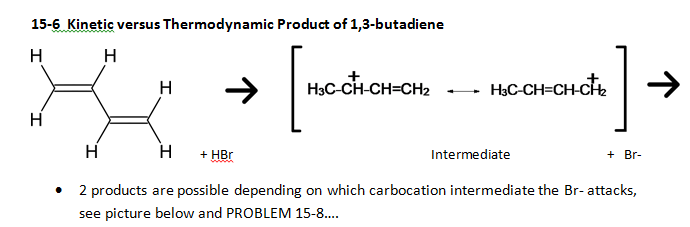
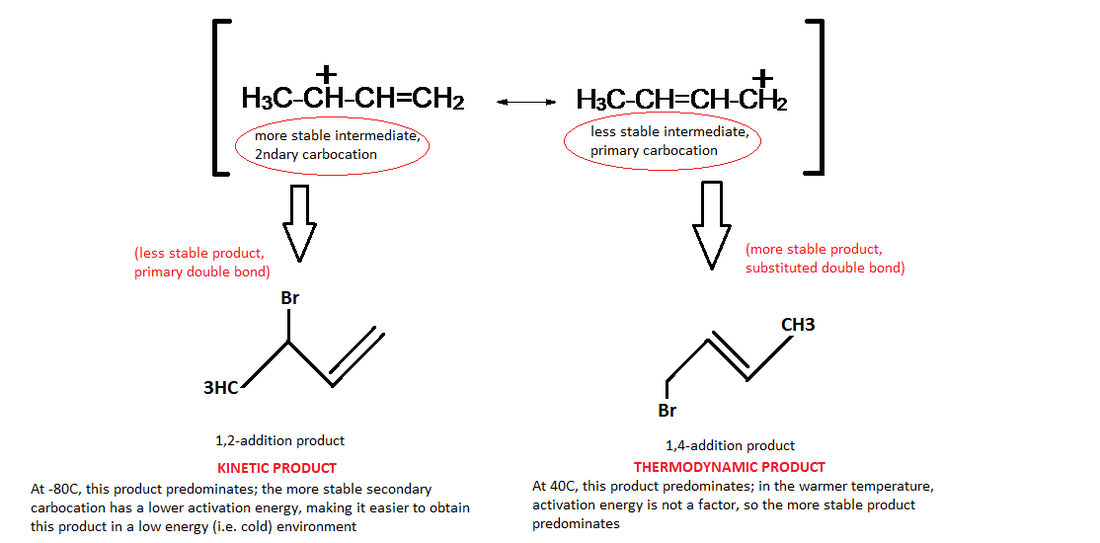
15-5: 1,2 and 1,4 addition to conjugated dienes [MECHANISM 15-1]
· Because of resonance, hydrogen halides (such as HBr) added across one of the double bonds of a diene can result in two different products, depending on which C the positive charge is on after the H is added.
· Allylic radicals are stabilized by resonance delocalization and can give multiple products
· Review the mechanism of radical reactions – there are three steps – initiation, propagation and termination. Here there are 2 propagation steps because there are two possible resonance intermediates
o Initiation
o Propagation step 1
o Propagation step 2
o Termination
o Regeneration of Br2
· Note that at high concentration adds across double bonds – if one has double bonds in the substrate, to avoid this, a low concentration of bromine is needed – and NBS is used
· N-bromosuccinimide (NBS) reacts with HBr to regenerate Br2 – keeps bromine concentration low by forming just enough bromine to keep the reaction going
· See problem 15-9, 15-10, 15-11
[Skipping 15-8, 15-9, 15-10, as they are not on the test]*Must confirm w/ Dr. Saadein for this year.
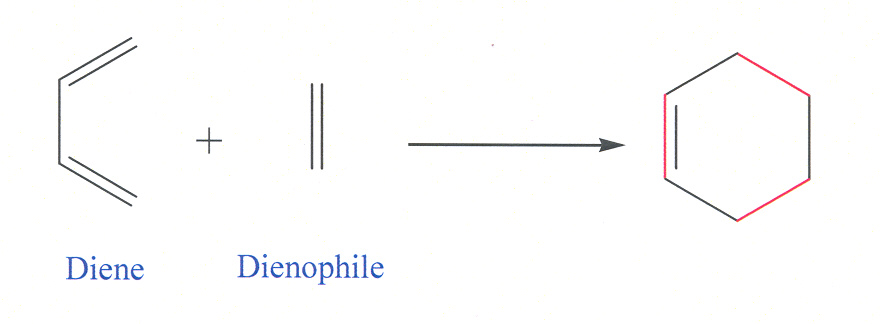
15-11 Diels-Alder Reaction [MECHANISM 15-3]
· For this section, understand the mechanism of and be able to draw Diels-Alder reaction products
· Diene reacts in the s-cis conformation, not s-trans
· What is s-cis versus s-trans? (end section 15-3)
· Electron adding groups can be placed on the diene and electron withdrawing groups can be placed on the dienophile to make them more reactive
· Some problems make require you to reconstruct the original diene/dienophile from a Diels-Alder product
· Problem Tip: A Diels-Alder product always contains one more ring than the reactants. The two ends of the diene form new bonds ot the ends of the dienophile. The center bond of the diene becomes a double bond. The dienophile’s double bond becomes a single bond (or its triple bond becomes a double bond).
· Problem Tip: To deconstruct a Diels-Alder product, look for the double bond at the center of what was the diene. Directly across the ring is the dienophile bond, usually with electron-withdrawing groups. (If a single bond, the dienophile had a double bond; if double, the dienophile had a triple bond.) Break the two bonds that join the diene and dienophile and restore the two double bonds of the diene and the double (or triple) bond of the dienophile.
· See problems 15-14, 15-15
· The Endo Rule: the endo position (dienophile part of the ring in the endo position, H-bonds in the exo position) is most stable, due to secondary overlap of the p-orbitals in the double bonds of each ring
· Remember that the 1,3-product is forbidden, due to impossible bonding/resonance patterns; 1,2 and 1,4-products are typically produced
· See problems 15-17, 15-18
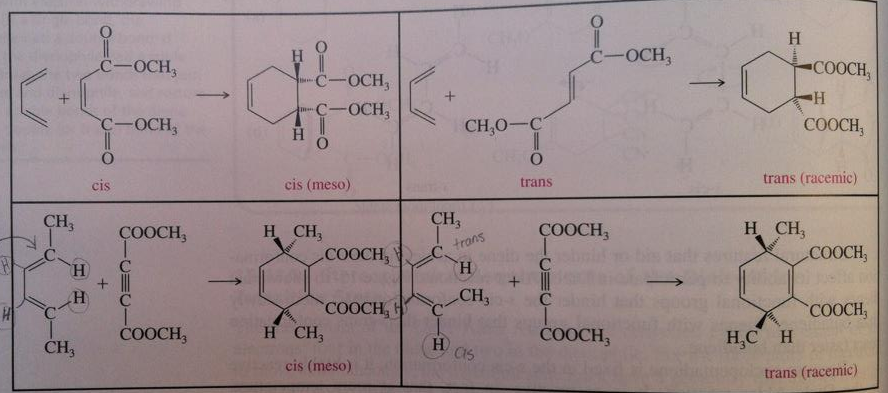
· Note that the diene in the top two squares does not change and the dienophile in the bottom two squares does not change
· The chart is saying that (on top) if you have a dienophile in cis or in trans, this will affect which way the bonds are pointing in the product
o Cis dienophile gives cis (meso) product
o Trans dienophile gives trans (racemic) product – because either group could be pointed up or down, as long as they are on opposite sides of each other
· (on bottom squares) If the diene has symmetrical substituents (left) it gives a cis(meso) product
· (on bottom squares) If the diene has asymmetrical substituents (right) it gives a trans (racemic) product
Key Points from Chapter 15-12: The Diels-Alder as an Example of Pericyclic Reaction
*Note: When doing problems dealing with molecular orbitals, remember the following…
Molecular Orbital Basics
· Molecular orbitals show all the different bonding and anti-bonding interactions that can exist between the molecule’s p orbitals in their double bonds.
· HOMO = highest occupied molecular orbital
· LUMO = lowest unoccupied molecular orbital
· HOMO* = new highest occupied molecular orbital in the molecule’s excited state
· A molecule will have twice as many p orbitals as it has double bonds; two p orbitals = one double bond
· The molecule has the same # of MOs as it has # of p orbitals
· Each p orbital contains one electron
· Thus, a MO will have as many electrons to fill it as it has p orbitals
· Electrons will always fill the lowest energy MO first, then the next lowest, etc.
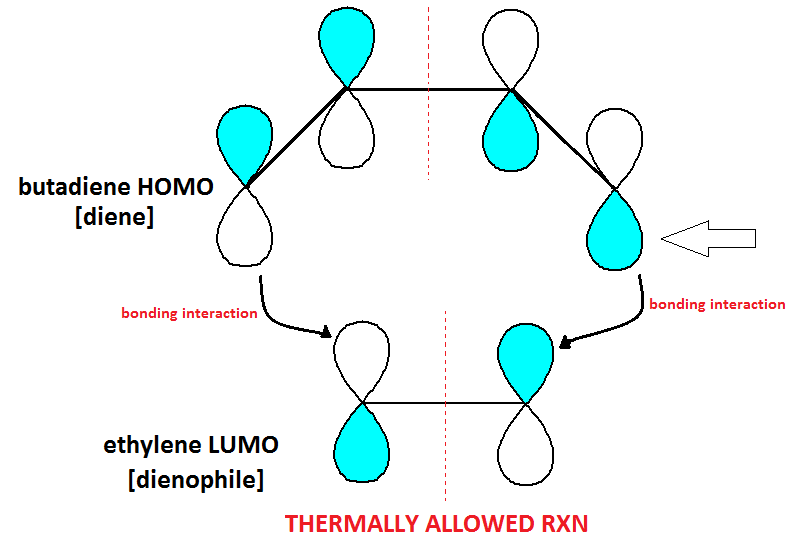
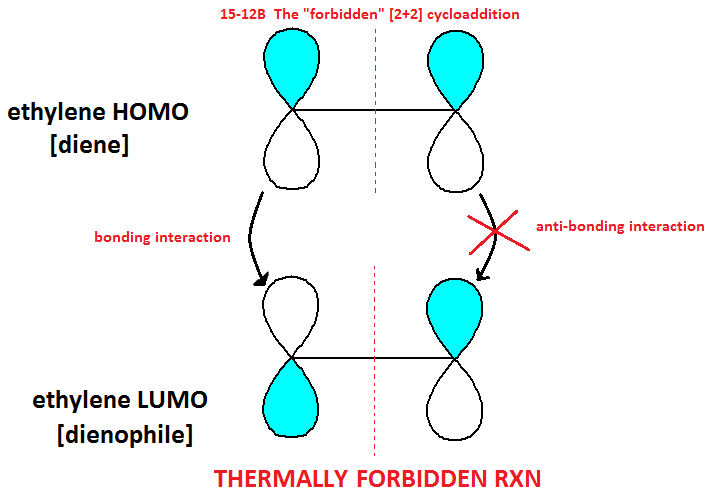
Determining Forbidden and Allowed Cycloadditions
· MOs can explain why some reactions are allowed and others are not.
· In a cycloaddition, the diene – or electron donator – will always donate electrons from its HOMO (the first electrons “off the top” so to speak).
· In a cycloaddition, the dienophile – or electron acceptor – will always accept electrons into its LUMO (first empty shell, essentially).
· When determining whether or not an addition reaction is allowed, keep in mind that the bottom half of the p orbitals in the diene always react with the top half of the p-orbitals in the dienophile (see diagram below)*SEE CLARIFICATION POST REGARDING THIS DIAGRAM.
· Thus, the LUMO is now occupied and in this brief transition state, it becomes the new HOMO, called HOMO*.
· HOMO* of an excited ethylene can donate its electron to a ground state ethylene LUMO, in an allowed reaction.
· In most cases, photochemically allowed reactions are thermally forbidden and thermally allowed reactions are photchemically forbidden.
o See problems 15-19, 15-20 and 15-35
Conjugation and UV absorption
· The energy difference between HOMO and LUMO decreases as the length of conjugation increases.
· A compound that contains a longer hcain of conjugated double bonds absorbs light at a longer wavelength (see the example of β-carotene in text).
· Only conjugated double bonds interact to give lower absorption frequencies. Isolated ones do not.
· An additional conjugated double bond increases the wavelength absorbed by ~30-40 nm, while an additional alkyl group increases it ~5 nm
o See problem 15-22



 RSS Feed
RSS Feed
