Experiments This Week
Monday – rtcB protein purification; Nanodrop revealed concentration too low (highest ~ 0.077 mg/ml). Started an overnight culture to try Shuman’s overnight expression protocol. Forgot to inoculate the culture with antibiotics.
Tuesday – Grew the culture with antibiotics until OD600 = 0.6. Expressed o/n at 19C.
Wednesday – rtcB protein purification; Nanodrop revealed concentration too low (highest ~ 0.081 mg/ml). I used new Ni:NTA beads. I suspect that no antibiotics in the overnight resulted in the culture ejecting the plasmid.
Thursday – Started an overnight culture with antibiotics (amp/km), to do a 1L Raines’s expression protocol on Friday.
Friday – Grew culture to OD600 = 0.6. Expressed rtcB protein for five hours. Harvested and froze pellets at -80C. Talked to Dr. Weinert and got a lot of good advice about future expressions.
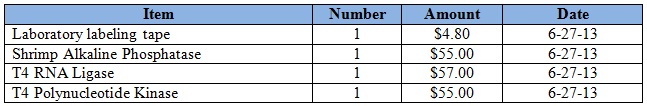
Purchases for the week = $171.80
Monday – preparing buffers, perhaps protein purification; meeting with Khalid at 4 p.m.
Tuesday – protein purification if I don't do it on Monday
Wednesday – setup of ligation reactions
Thursday – gels of ligation reactions; setup of additional reactions
Friday – other ligation gels; may attempt an overnight expression during the week, depending on how the gels go
Tips
Dr. Weinert has a cooled incubator (16-19C etc) for growing overnight expression cultures. Very cool. It’s in the same room as the scales for balancing samples.
Learned how to autoclave biohazard waste. Put it on #3 “solids” in the downstairs autoclave. Can change the time it cooks the samples on the autoclave – hit “menu” and use the arrow keys. In order to get it to start after that, have to hit “advanced,” and it’ll go back to normal.
Interestingly, using vacuum sealing grease on the inside of the gel plates did not stop the samples from leaking. They leaked faster than I’ve ever seen. However, I also used parafilm on the bottom and wonder if it was creating a bad seal. Need to check this more in depth. I’ll try subbing in Vaseline instead of vacuum sealing grease, so I don’t steal all of Dr. Weinert’s expensive grease.
There’s another 50 ml falcon tube centrifuge like we have in the cell culture room in the Lynn lab incubator room. Useful if ours is busy. The one that’s in the Lynn lab next to the UV-Vis is no longer working, for some reason.
Using silicone vacuum grease between the plates of the SDS-PAGE gels did not appear to work. They still leaked. Not sure if that’s from the sides or the bottom. Will have to check.
One can dissolve silicone vacuum grease off of glass with acetone. It seems to really mess up Rainex though.






 RSS Feed
RSS Feed
