“Oh – oxalic acid
My – malonic acid
Such – succinic acid
Good – glutaric acid
Apple – adipic acid
Pie!” – pimelic acid
*Know the IUPAC names of the compounds in table 20-1 and the common names of the first nine.
| IUPAC Name methanoic ethanoic propanoic 2-propenoic butanoic 2-methylpropanoic trans-2-butenoic pentanoic 3-methylbutanoic | Common Name formic acetic propionic acrylic butyric isobutyric crotonic valeric isovaleric |
lauric, myristic, palmitic, stearic – unsaturated fatty acids
oleic, linoleic – unsaturated fatty acids
Naming Tip: Note that “oxo” is the name of a C=O (carbonyl) when it appears as a group, while “-one” is the suffix name of a ketone. Carboxylic acids take precedence over carbonyl groups when it comes to the primary name of the compound. (Cmds with a C=O and a COOH will be named with the “-ic acid” suffix over “-one” of “ketone.”) Don’t mix those up!
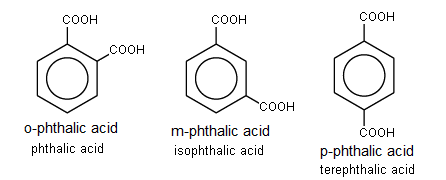
Note: Benzenoid cmds with two COOH groups are named phthalic acids
Examples:
1. COOH are more acidic than alcohols and dissociate more easily (due to spreading negative charge over two O’s, versus one in –OH.
2. Typical pKa’s of COOH are around ~5. Typical alcohol pKa’s are ~16-18.
3. For dicarboxylic acids, the second COOH group is always less acidic than the first (Ka2 << Ka1), because extra energy is required to create a 2nd (-) charge close to another, mutually repulsive (-) charge.
4. Electronegative atoms enhance the strength of a COOH in their vicinity by stabilizing the (-) charged carboxylate ion through an inductive e- withdrawing effect and increasing its acidity. This effect is especially noticeable when the substituent is on the alpha carbon.
5. Electron donating group have the opposite effect, destabilizing the COOH and decreasing its acidity.
6. The donating or withdrawing tendencies of groups are strongest when they are on the ortho or para positions.
Salt of a carboxylic acid: Combination of a carboxylate ion and a cation
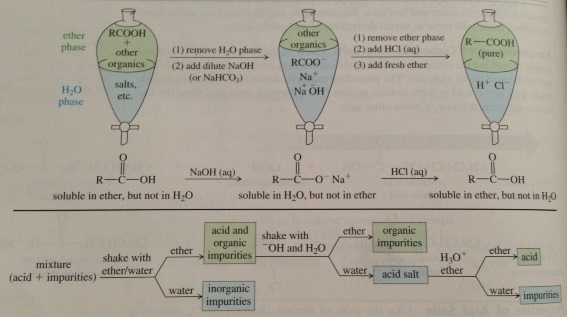
Acid/Base Extraction
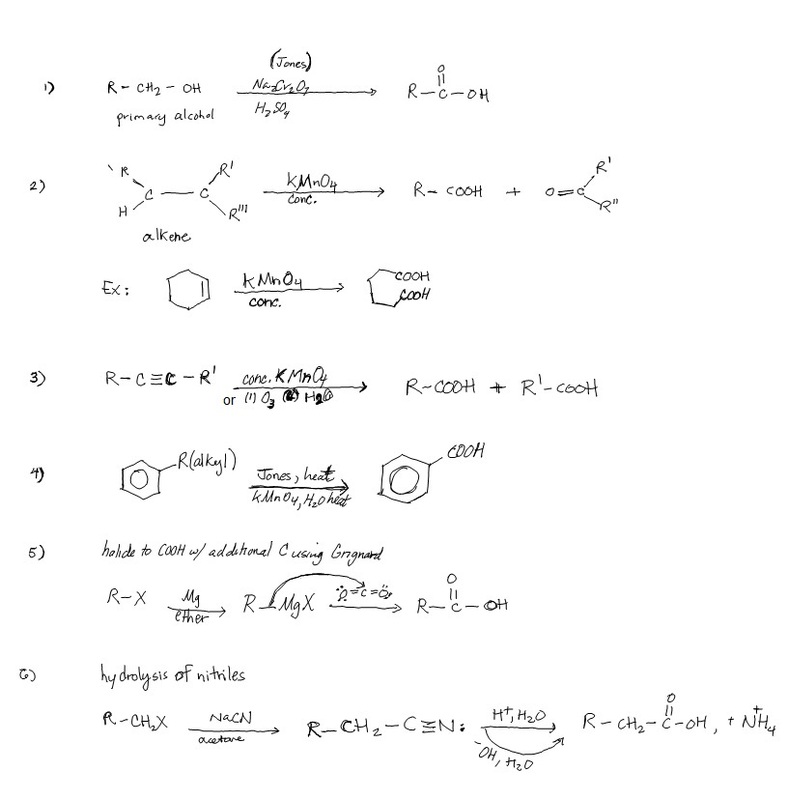
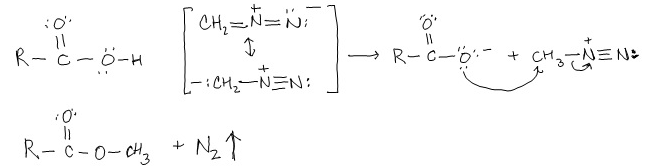
Synthesis of COOHs
Previously
Interconverting between these derivatives involves nucleophilic acyl substitution and can be grouped under whether the reaction occurs in …
a. Acid
b. Base
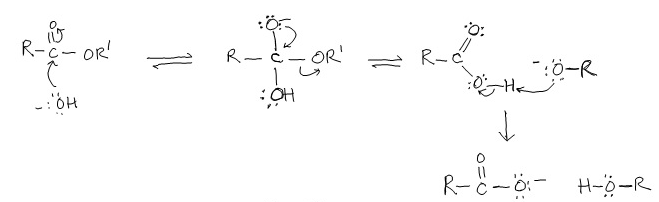
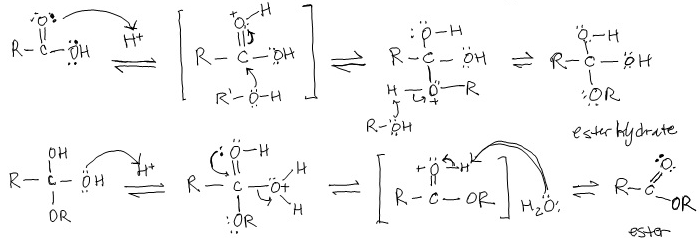
Nucleophilic Acyl Substitution (in base)
Under basic conditions, a strong nucleophile adds to the carbonyl group, creating a tetrahedral intermediate that then expels the leaving group (which one is already there). If an alkoxide ion is created, it quickly (immediately) reacts with the COOH to create an alcohol and a carboxylate ion.
Example:
Condensation of COOH w/ Amines to Produce Amides
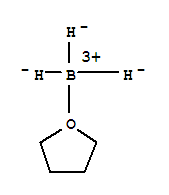
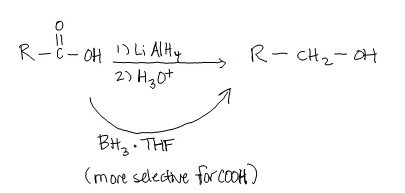
There are two major ways the book discusses reducing COOH to a primary alcohol:
1) Using lithium hydride and water
2) Using borane complexed with THF (more selective)
Be aware of the mechanism by which LiAlH4 reduces COOH
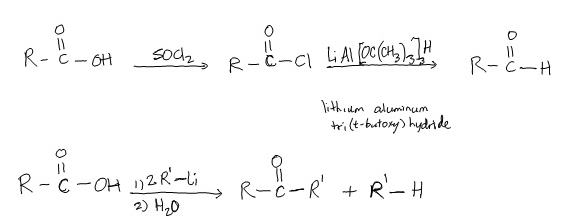
Acids chlorides are an easily made and reactive derivative of a COOH that is often used to form the other derivatives. The carbonyl oxygen and the chlorine withdraw e density from the carbonyl C making it strongly electrophilic.
Thus, acid chlorides are widely used in nucleophilic acyl substitution reactions, following the addition-elimination mechanism shown above.

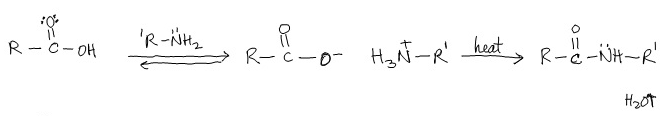

 RSS Feed
RSS Feed
